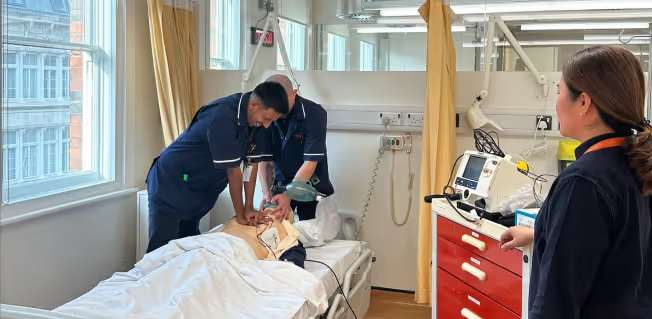
Immediate Life Support & e-Immediate Life Support Courses
Equips healthcare professionals with essential skills to manage cardiac arrest and deteriorating patients, enhancing their capabilities as first responders and team leaders in emergencies.
Learning outcomes

Identify the causes and promote the prevention of cardiopulmonary arrest.

Recognise and treat the deteriorating patient using the ABCDE approach.

Utilise non-technical skills to facilitate team leadership and effective team membership.

Undertake the skills of quality CPR and defibrillation (manual and/or AED) and simple airway manoeuvres;
Formats available
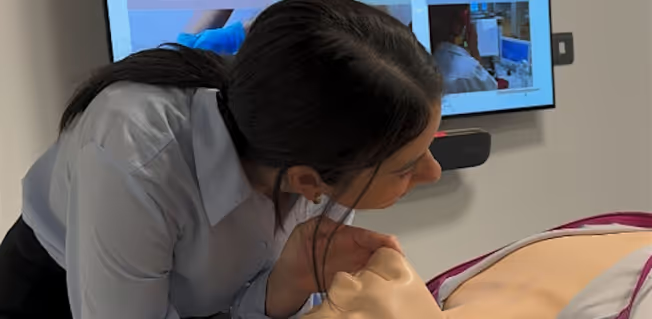
Basic Life Support level 2 (BLS)
A two-hour training for health and social care workers, teaching essential emergency skills like CPR, AED use, choking management, and the recovery position.
Learning outcomes

Awareness of RCUK 2021 guidelines and Chain of Survival.

Demonstrated CPR and the safe use of AED as per RCUK 2021 guidelines.

Awareness of choking management in adults.

Awareness of recovery position.
Formats available


Cannulation Course (coming soon)
This 4-hour course provides participants with essential knowledge and practical skills in performing cannulation. Designed for healthcare professionals seeking to enhance their competency in this technique, the course includes hands-on practice and instruction.
Upon completion, participants will receive a certificate of participation, acknowledging their engagement in the training. Please note, this certificate does not confer formal certification or qualification in cannulation.

Ultrasound Guided cannulation Course (coming soon)
This 4-hour course provides healthcare professionals with the essential skills and knowledge required to perform ultrasound-guided cannulation. The course combines theoretical instruction with hands-on practice, ensuring participants gain practical experience in using ultrasound technology to assist with cannulation procedures.
Upon completion, participants will receive a certificate of participation, acknowledging their engagement in the training. Please note, this certificate does not confer formal certification or qualification in ultrasound-guided cannulation.















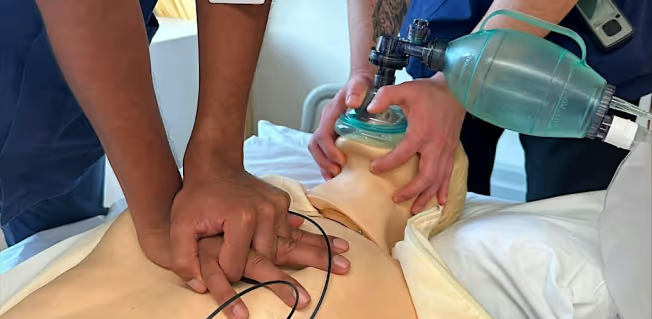





.avif)
